-
摘要:
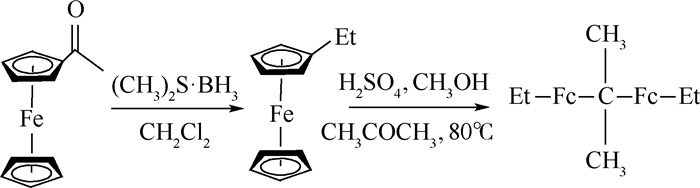
2,2'-双(乙基二茂铁)丙烷(卡托辛)是一种综合性能优良的高效液体燃速催化剂,主要包含4种乙基取代位置不同、物理和化学性质均很相近的卡托辛同分异构体和不同取代基的双二茂铁丙烷,其中卡托辛同分异构体与其他双二茂铁丙烷很难分离。首先,采用密度泛函理论(DFT)方法模拟确定了4种同分异构体的结构,分别获得4种同分异构体的1H-NMR和13C-NMR理论谱图。其次,计算研究了合成过程中温度对产物中4种同分异构体含量的影响。最后,在理论合成条件的指导下,以高纯度乙基二茂铁和丙酮为原料、浓硫酸为催化剂,合成了卡托辛粗品。结果表明:利用惰性气体辅助蒸汽蒸馏法得到纯度大于99.0%的卡托辛同分异构体,收率高于94.0%,并通过核磁共振法确定了其结构。
-
关键词:
- 2, 2′-双(乙基二茂铁)丙烷 /
- 合成 /
- 提纯 /
- 高含量 /
- 密度泛函理论(DFT)
Abstract:2, 2'-bis (ethylferrocenyl) propane (catocene) is a kind of high-efficiency burning rate liquid catalyst with excellent comprehensive performance. It is a mixture with four isomers of the different substituting positions of ethyl groups and other different substitutions of diferrocenyl propane compounds, which are difficult to separate due to their similarly physical and chemical properties. In this paper, Density Functional Theory (DFT) was used to simulate and determine the structure of the four isomers, and 1H-NMR and 13C-NMR spectra of the four isomers were obtained respectively. Meanwhile, the influence of reaction temperature on the content of four isomers in the product was studied. Under the guidance of the theory, the crude catocene was synthesized from high-purity ethylferrocene and acetone with sulfuric acid as catalyst and was purified by vapor distillation in inert gas atmosphere. The yield of catocene is more than 94.0% with a purity of more than 99.0%. The structure of synthetic product is confirmed by nuclear magnetic resonance spectra.
-
表 1 蒸馏溶剂配比对产品的影响
Table 1. Influence of the ratio of solvent on distillation effect of product
水-乙醇体积比 收率/% 卡托辛同分异构体相对含量/% 95∶5 82 97.2 93∶7 89 99.0 92∶8 95 99.4 90∶12 82 99.2 88∶14 71 99.3 表 2 温度对卡托辛同分异构体蒸馏效果的影响
Table 2. Influence of temperatures on distillation effect of catocene isomers
蒸馏瓶温度/℃ 蒸汽发生器温度/℃ 卡托辛同分异构体相对含量/% 收率/% 120 190 97.35 96 125 190 98.83 96 130 190 99.22 95 135 190 99.26 90 140 190 98.74 81 130 170 98.81 93 130 180 99.01 92 130 200 99.12 87 -
[1] USMAN M, WANG L, YU H, et al. Recent progress on ferrocene-based burning rate catalysts for propellant applications [J]. Journal of Organometallic Chemistry, 2018, 872: 40-53. doi: 10.1016/j.jorganchem.2018.07.015 [2] ZAIN-UL-ABDIN, WANG L, YU H, et al. Tris (2-aminoethyl) amine-based ferrocene-terminated dendrimers as burning rate catalysts for ammonium perchlorate-based propellant decomposition[J]. Applied Organometallic Chemistry, 2018, 32(4): 264-270. doi: 10.1002/aoc.4268 [3] KAI F G, HANSEN F K. Diffusion rates and the role of diffusion in solid propellant rocket moto radhesion[J]. Journal of Applied Polymer Science, 2010, 103(3): 1529-1538. http://www.onacademic.com/detail/journal_1000033761356710_f383.html [4] ZHAO H, JIN Z, SU H, et al. Targeted synthesis of a 2D ordered porous organic framework for drug release[J]. Chemical Communications, 2011, 47(22): 6389-6391. doi: 10.1039/c1cc00084e [5] STERESCU D M, STAMATIALIS D F, MENDES E, et al. Boltorn-modified poly (2, 6-dimethyl-1, 4-phenylene oxide) gas separation membranes[J]. Macromolecules, 2007, 40(15): 5400-5410. doi: 10.1021/ma070772g [6] GULDI D M, LUO C P, SWARTZ A, et al. π-conjugated electroactive oligomers: Energy and electron transducing systems[J]. Journal of Physical Chemistry, 2004, 108(3): 455-467. doi: 10.1021/jp034186a [7] JIANG J X, TREWIN A, ADAMS D J, et al. Band gap engineering in fluorescent conjugated microporous polymers[J]. Chemical Science, 2011, 2(9): 1777-1781. doi: 10.1039/c1sc00329a [8] LI S C, ZHENG Y, GAO F, et al. Experimental and computational interrogation of fast SCR mechanism and active sites on H-Form SSZ-13[J]. ACS Catalysis, 2017, 7(8): 5087-5096. doi: 10.1021/acscatal.7b01319 [9] KHAMRANG T, VELUSAMY M, MADHAVAN J, et al. A combined experimental and computational investigations on pyrene based D-π-A dyes[J]. Physical Chemistry Chemical Physics, 2018, 20(9): 6264-6273. doi: 10.1039/C7CP08038G [10] HIROTO T, KAZUKO H, SADAYUKI W, et al. Local structures and electronic states of C-S-H-Sodium-H2O interface: NMR and DFT studies[J]. Journal of Physical Chemistry, 2020, 124(10): 5672-5680. doi: 10.1021/acs.jpcc.9b11302 [11] MALENOV D P, NINKOVIC D B, ZARIC S D. Stacking of metal chelates with benzene: Can dispersion-corrected DFT be used to calculate organic-inorganic stacking [J]. Chemphyschem, 2015, 16(4): 761-768. doi: 10.1002/cphc.201402589 [12] ZHOU W J, LIU J B, STEVEN D, et al. Molecular dynamics simulations, reaction pathway and mechanism dissection, and kinetics modeling of the nitric acid oxidation of dicyanamide and dicyanoborohydride anions[J]. Journal of Physical Chemistry, 2020, 124(49): 11175-11188. doi: 10.1021/acs.jpcb.0c07823 [13] YANG Z, SEBASTIAN T. Binuclear Pd(Ⅰ)-Pd(Ⅰ) catalysis assisted by iodide ligands for selective hydroformylation of alkenes and alkynes[J]. Journal of the American Chemical Society, 2020, 142(42): 18251-18265. doi: 10.1021/jacs.0c09254 [14] SABER M, MAXIM V I, QADIR K T. Improving performance of the SMD solvation model: Bondi radii improve predicted aqueous solvation free energies of ions and pKa values of thiols[J]. Journal of Physical Chemistry, 2019, 123(44): 9498-9504. doi: 10.1021/acs.jpca.9b02340 [15] RAO Z Z, MASAYOSHI T, MASATAKA N. Ab initio quantitative prediction of tacticity in radical polymerization of poly(methyl methacrylate) by a molecular simulation technique with the conformation indexing for multiple transition states[J]. Journal of Physical Chemistry, 2020, 124(31): 16895-16901. doi: 10.1021/acs.jpcc.0c01812 -






 下载:
下载: