-
摘要:
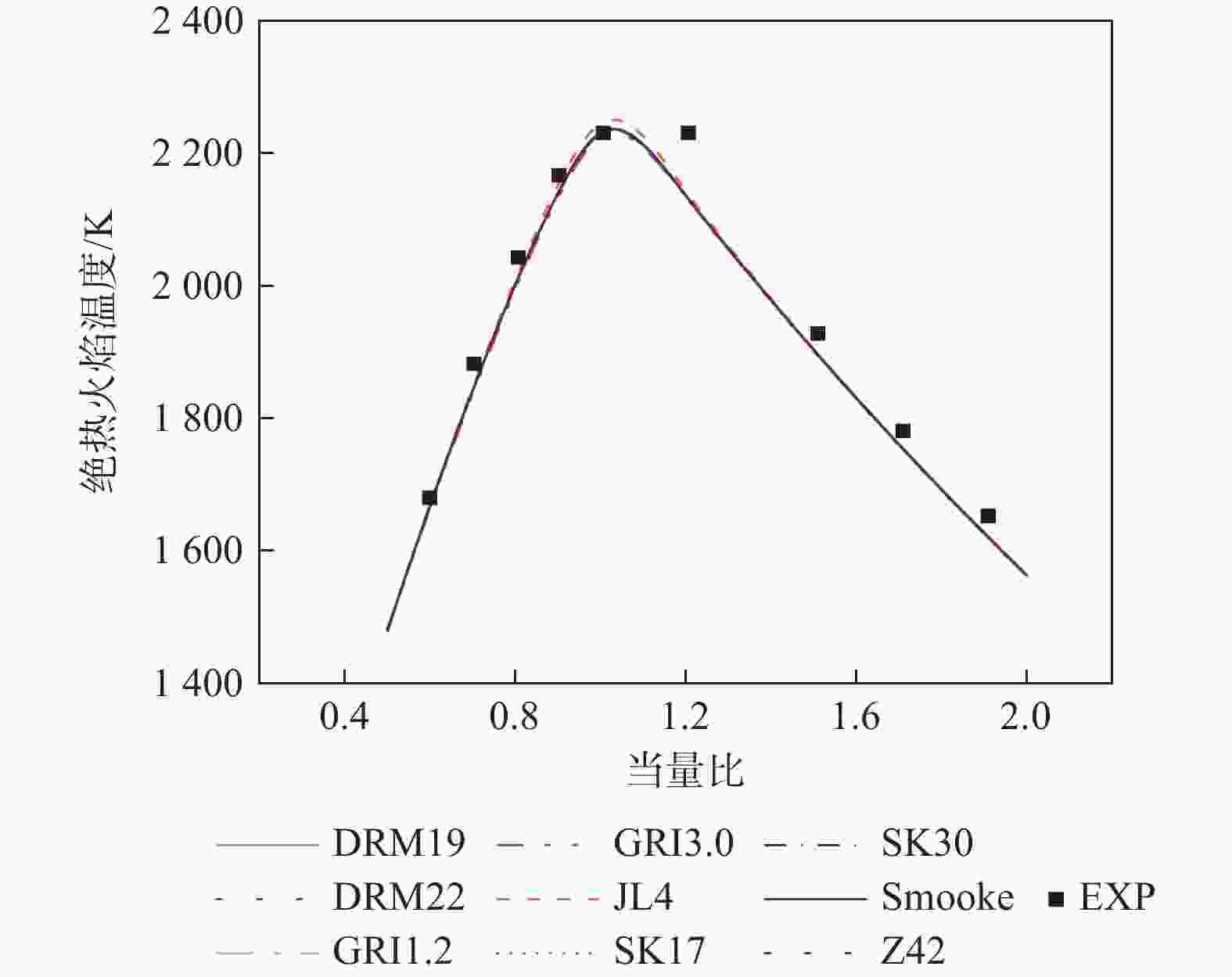
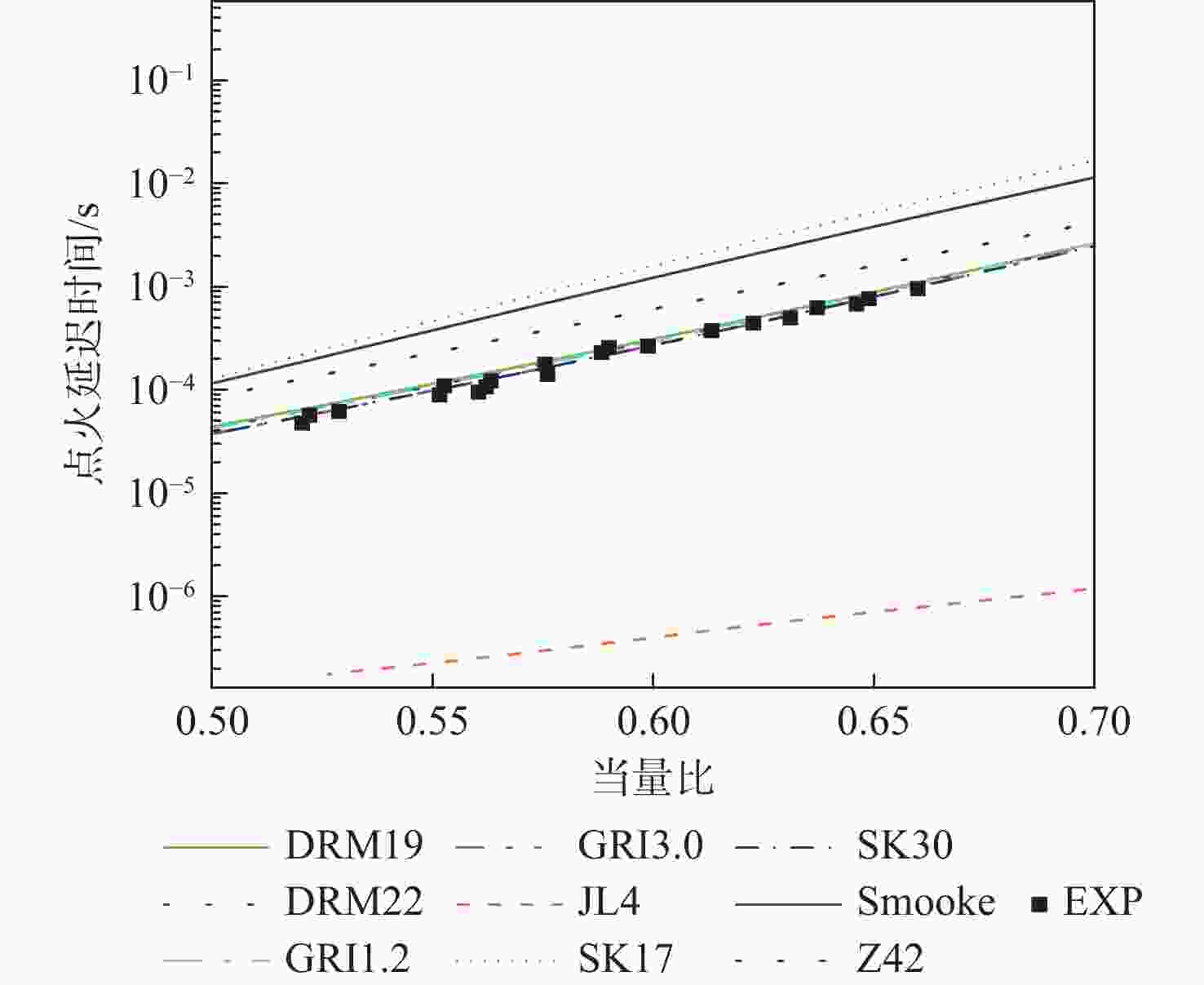
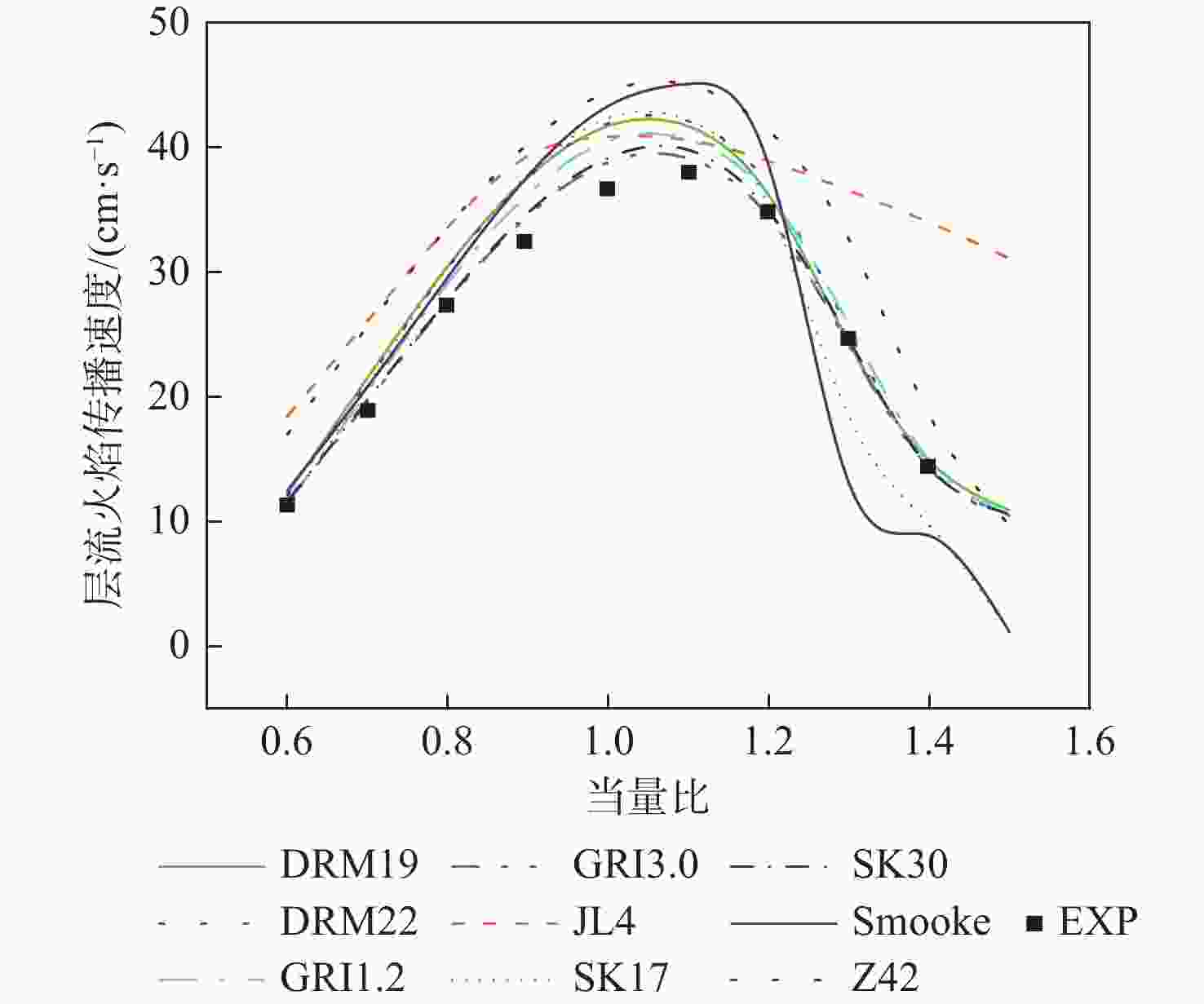
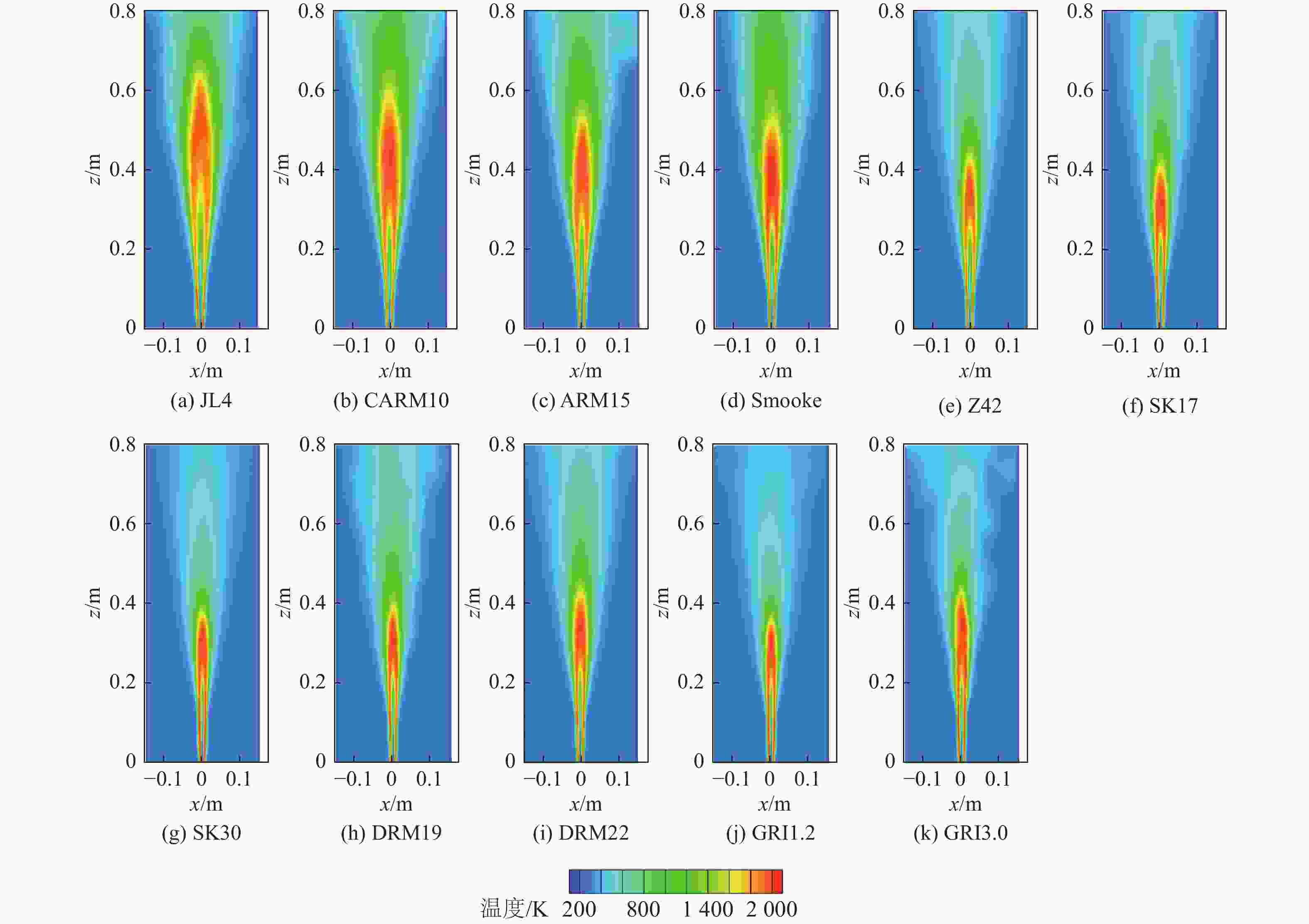
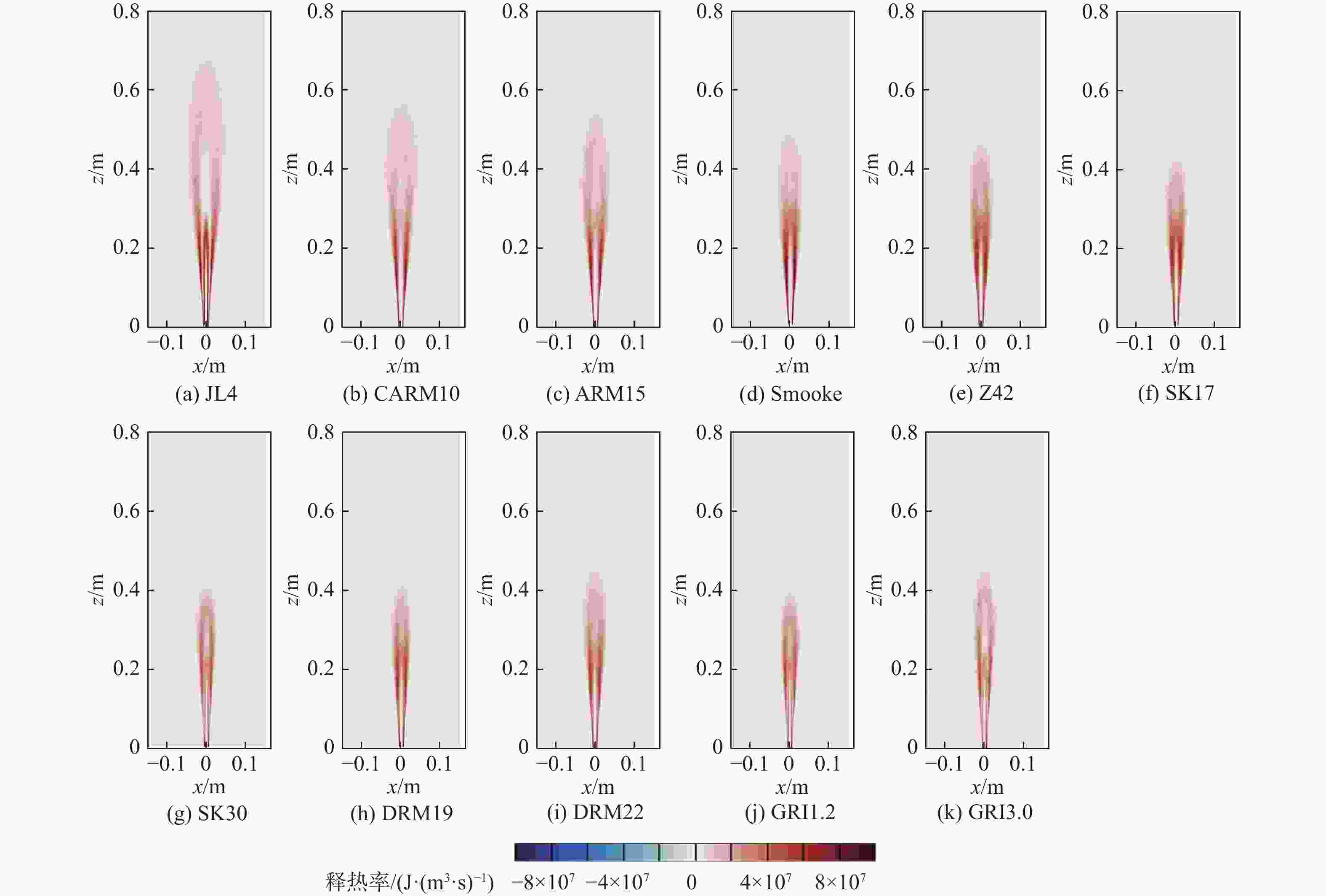
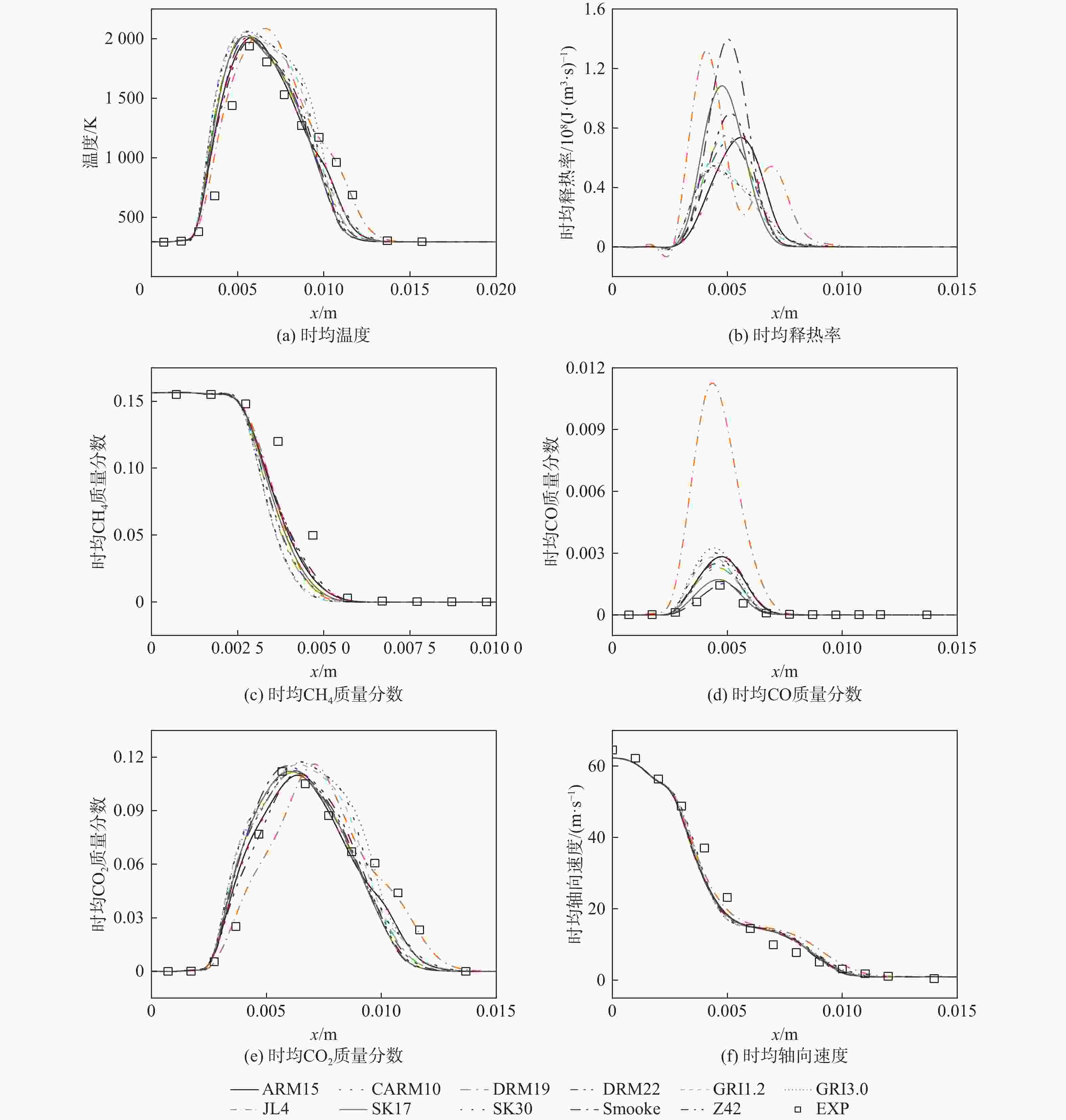
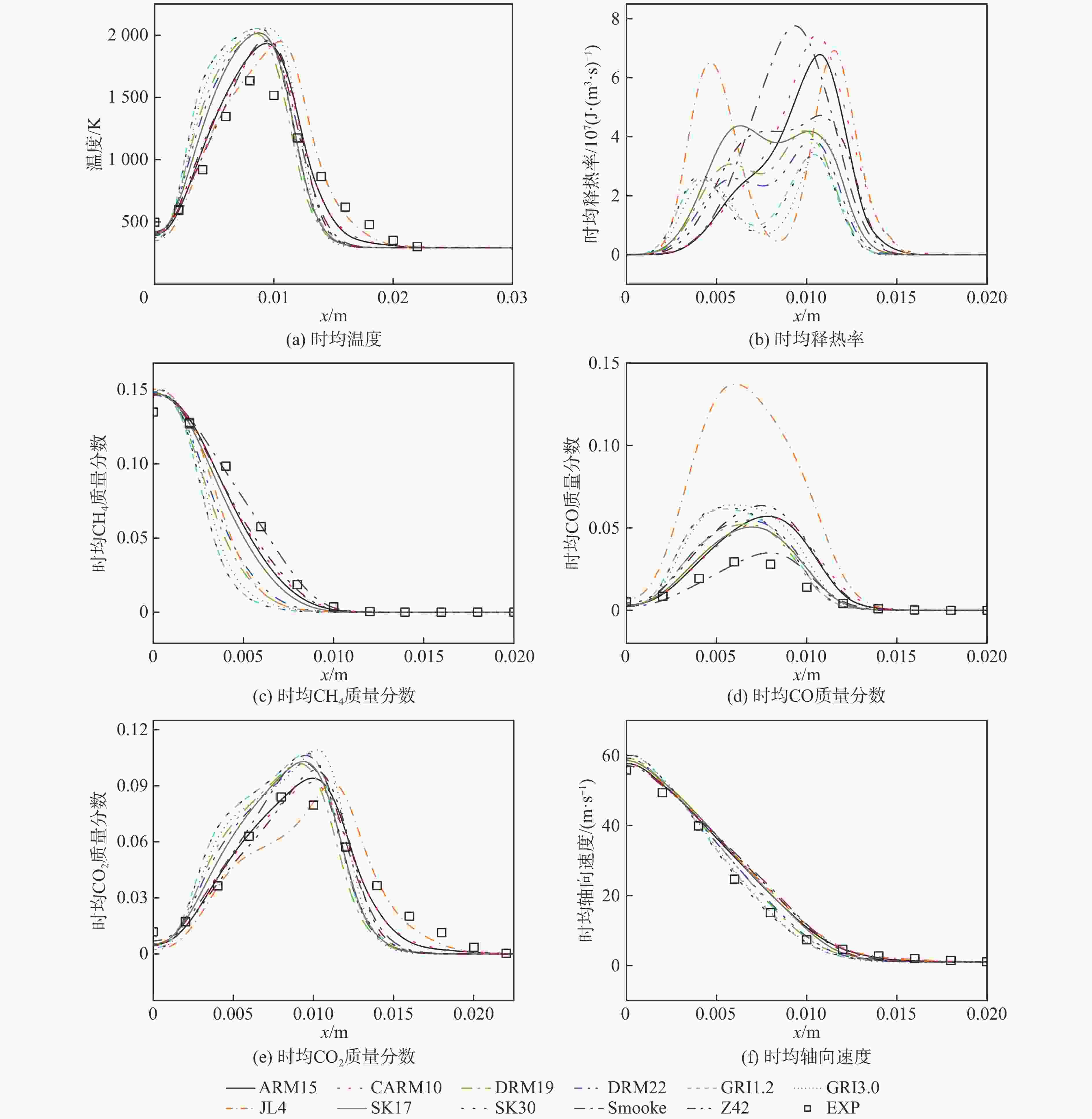
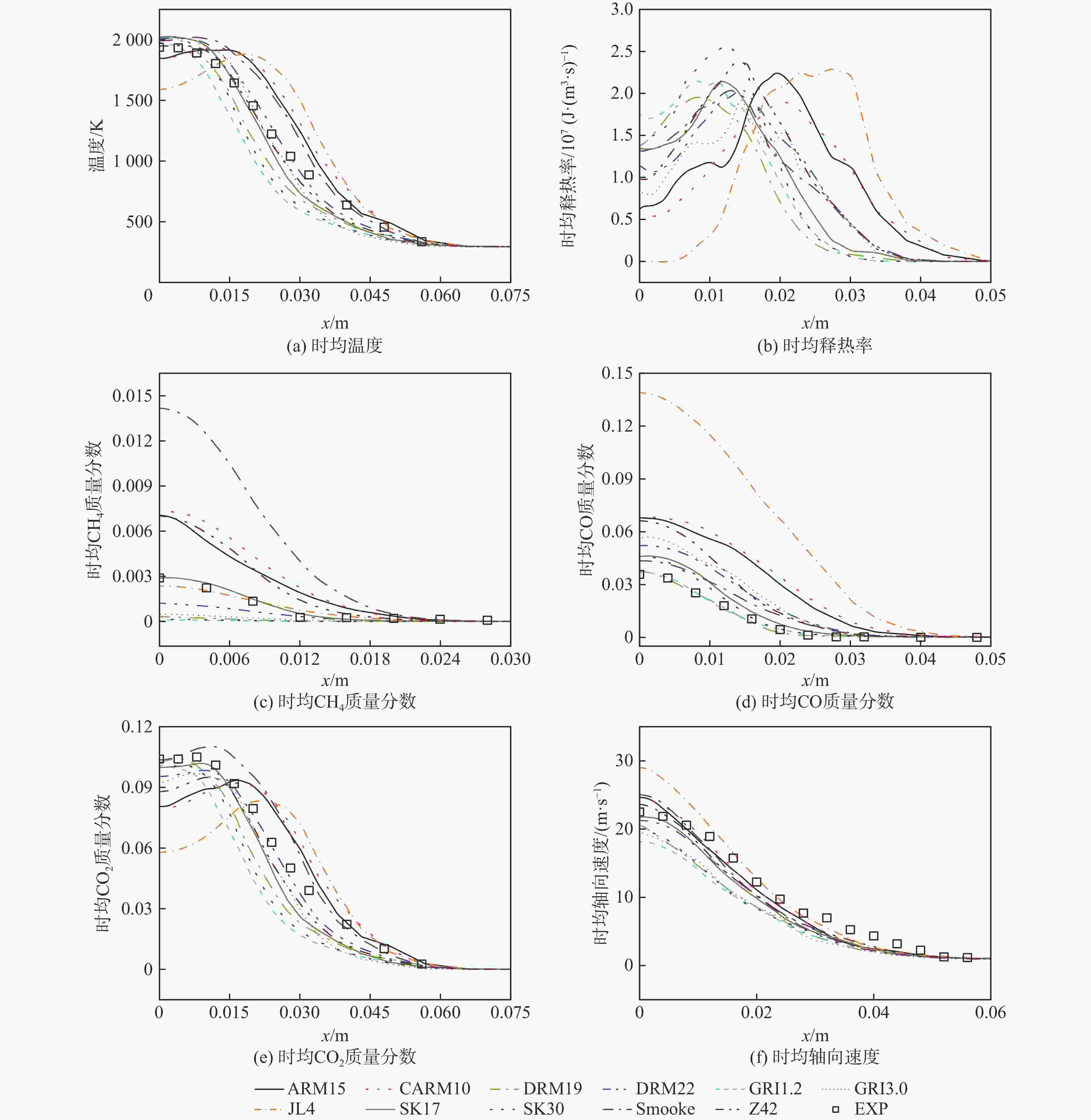
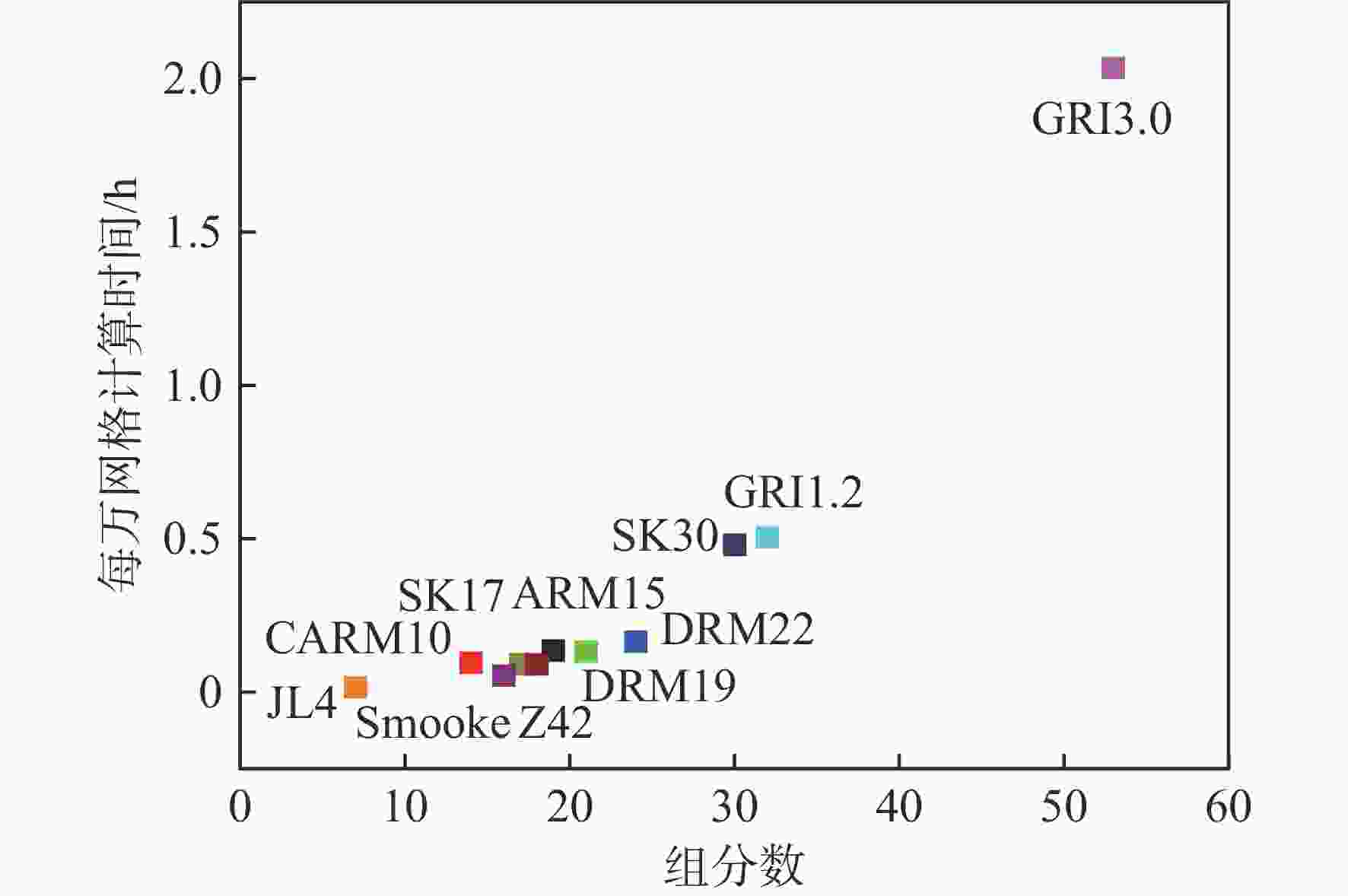
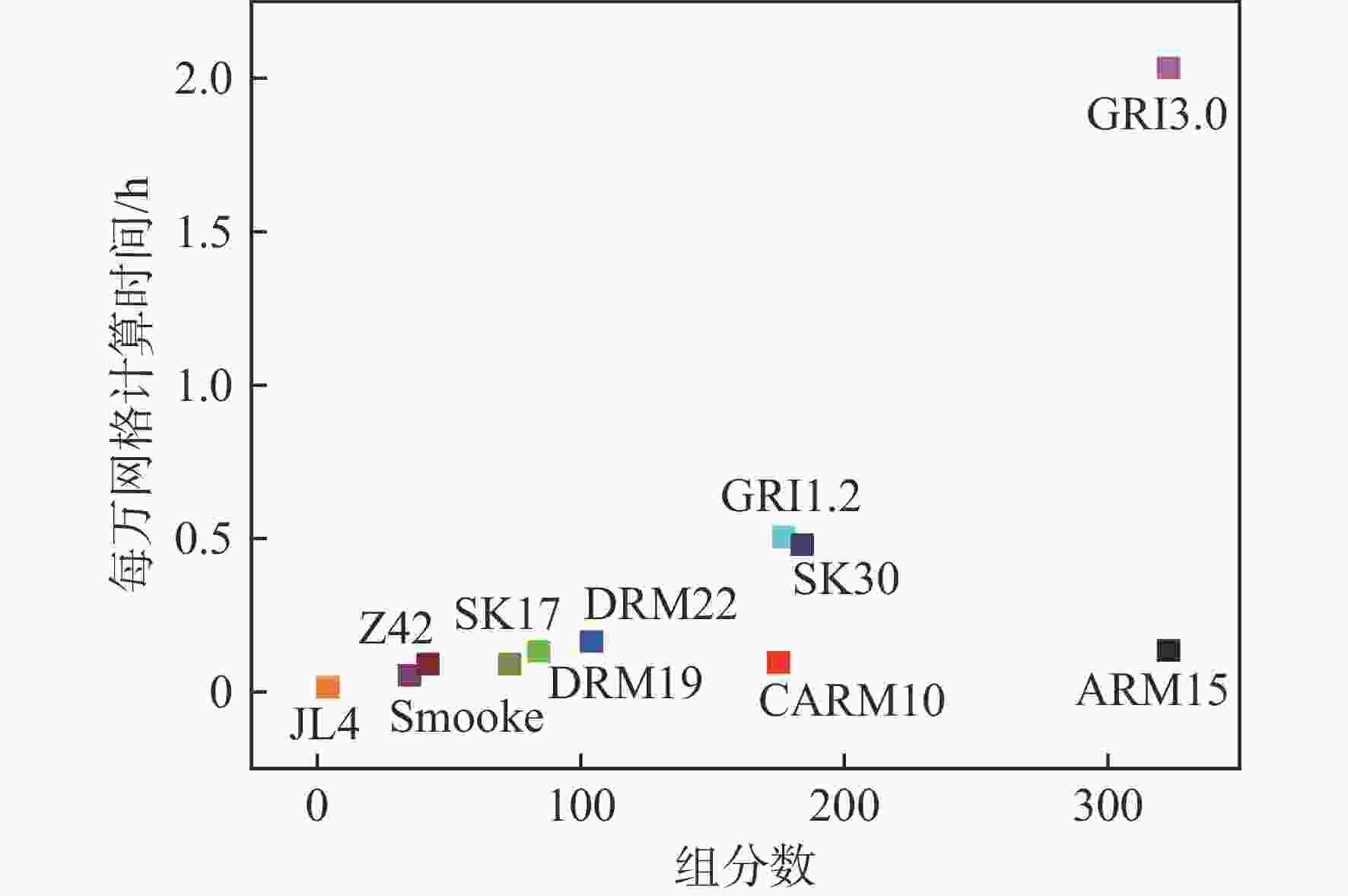
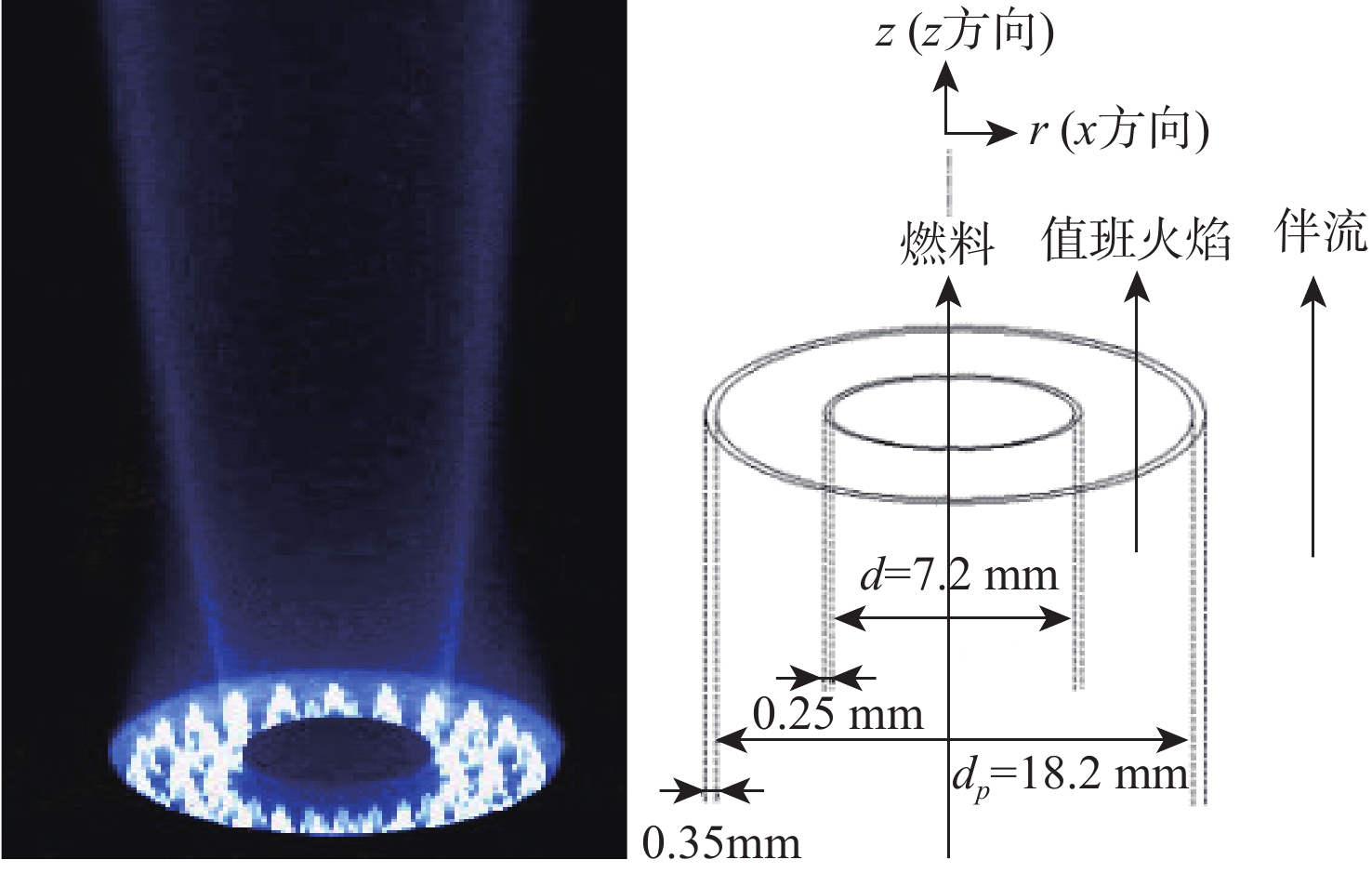
湍流燃烧在在工业与运输业中的应用广泛,燃烧过程中流动与化学反应强烈耦合,且耦合机理复杂,因而燃烧模拟中需要引入化学反应机理。化学反应机理种类多、数量大,且机理的选择对于模拟结果的影响巨大。为了探究化学反应机理对于湍流射流火焰模拟的影响,选择了11种不同的化学反应机理,对其进行了反应动力学分析,选择Sandia Flame D湍流射流火焰作为研究对象,提取了计算稳定后的温度、组分浓度等数据进行对比分析。结果表明:不同化学机理的动力学表现差异较大,其中,总包机理JL4由于省略了过多的中间产物,导致点火延迟时间过短,而SMOOKE和z42机理由于省略了多碳原子组分,导致点火延迟时间较实验值更长;不同机理的计算结果随流场的发展而差异增大,靠近燃料进口处的差异主要来源于动力学表现的差异,而远离燃料进口处的差异则更多来源于不同化学反应机理中参与的组分、反应种类不同;对于所有的化学机理,存在当中使用的组分数越多,化学反应的高释热率区域越集中,火焰长度越短,全场最高温度越高的趋势。
-
关键词:
- 化学反应机理 /
- 大涡模拟-运输概率密度函数 /
- 湍流火焰 /
- 燃烧仿真 /
- Sandia Flame D涡流射流火焰
Abstract:Turbulent combustion is extensively employed in industry and transportation, where the flow and chemical reactions are intricately interconnected. Due to the complexity of this coupling mechanism, it is essential to incorporate the chemical reaction mechanism in combustion simulations. However, there are many kinds and quantities of chemical reaction mechanisms, and the choice of mechanism has great influence on the simulation results. In this article, 11 distinct chemical reaction mechanisms were chosen and their reaction kinetics examined in order to investigate the impact of chemical reaction mechanisms on the simulation of turbulent jet flame. Then Sandia Flame D turbulent jet flame was selected as the research object, and the temperature and component concentration data after calculation and stability were extracted for comparative analysis. The findings demonstrate the stark differences in the kinetic performance of several chemical processes. The ignition delay time of the global mechanism JL4 is too short due to the omission of too many intermediate species. The ignition delay time of the SMOOKE mechanism and the z42 mechanism is longer than the experimental value due to the omission of multi-carbon species. As the flow field develops, the disparity between the computation outputs of various methods grows. Whereas the difference away from the fuel inlet is mostly caused by the various species and elementary processes involved in various chemical reaction mechanisms, the difference near the fuel inlet is primarily caused by the difference in kinetic performance. For all chemical mechanisms, there is a tendency that the more species used in the chemical reaction, the more concentrated the region of high heat release rate, the shorter the flame length, and the higher the maximum temperature throughout the field.
-
表 1 本文使用的甲烷化学反应机理
Table 1. The chemical reaction mechanism of methane used in this paper
种类 JL4 SMOOKE Z42 sk17 sk30 DRM19 DRM22 GRI1.2 GRI3.0 求和 CH4 1 1 1 1 1 1 1 1 1 9 CO 1 1 1 1 1 1 1 1 1 9 CO2 1 1 1 1 1 1 1 1 1 9 H2 1 1 1 1 1 1 1 1 1 9 H2O 1 1 1 1 1 1 1 1 1 9 N2 1 1 1 1 1 1 1 1 1 9 O2 1 1 1 1 1 1 1 1 1 9 CH2O 0 1 1 1 1 1 1 1 1 8 CH3 0 1 1 1 1 1 1 1 1 8 H 0 1 1 1 1 1 1 1 1 8 HCO 0 1 1 1 1 1 1 1 1 8 HO2 0 1 1 1 1 1 1 1 1 8 O 0 1 1 1 1 1 1 1 1 8 OH 0 1 1 1 1 1 1 1 1 8 CH2 0 0 1 1 1 1 1 1 1 7 CH3O 0 1 1 0 1 1 1 1 1 7 CH2(S) 0 0 0 1 1 1 1 1 1 6 H2O2 0 1 1 0 1 0 1 1 1 6 C2H4 0 0 0 0 1 1 1 1 1 5 C2H5 0 0 0 0 1 1 1 1 1 5 C2H6 0 0 0 0 1 1 1 1 1 5 AR 0 0 0 0 0 1 1 1 1 4 C2H2 0 0 0 0 1 0 1 1 1 4 C2H3 0 0 0 0 1 0 1 1 1 4 CH 0 0 1 0 1 0 0 1 1 4 CH2OH 0 0 0 1 1 0 0 1 1 4 C 0 0 0 0 1 0 0 1 1 3 CH2CO 0 0 0 0 1 0 0 1 1 3 CH3OH 0 0 0 0 1 0 0 1 1 3 HCCO 0 0 0 0 1 0 0 1 1 3 C2H 0 0 0 0 0 0 0 1 1 2 CH2CHO 0 0 0 0 1 0 0 0 1 2 HCCOH 0 0 0 0 0 0 0 1 1 2 C3H7 0 0 0 0 0 0 0 0 1 1 C3H8 0 0 0 0 0 0 0 0 1 1 CH3CHO 0 0 0 0 0 0 0 0 1 1 CN 0 0 0 0 0 0 0 0 1 1 H2CN 0 0 0 0 0 0 0 0 1 1 HCN 0 0 0 0 0 0 0 0 1 1 HCNN 0 0 0 0 0 0 0 0 1 1 HCNO 0 0 0 0 0 0 0 0 1 1 HNCO 0 0 0 0 0 0 0 0 1 1 HNO 0 0 0 0 0 0 0 0 1 1 HOCN 0 0 0 0 0 0 0 0 1 1 N 0 0 0 0 0 0 0 0 1 1 N2O 0 0 0 0 0 0 0 0 1 1 NCO 0 0 0 0 0 0 0 0 1 1 NH 0 0 0 0 0 0 0 0 1 1 NH2 0 0 0 0 0 0 0 0 1 1 NH3 0 0 0 0 0 0 0 0 1 1 NNH 0 0 0 0 0 0 0 0 1 1 NO 0 0 0 0 0 0 0 0 1 1 NO2 0 0 0 0 0 0 0 0 1 1 注:1为包含,0为不包含。 化学反应 JL4 SMOOKE Z42 sk17 sk30 DRM19 DRM22 GRI1.2 GRI3.0 求和 O+H2=H+OH 0 1 1 1 1 1 1 1 1 8 O+CH3=H+CH2O 0 1 1 1 1 1 1 1 1 8 H+O2+M=HO2+M 0 1 1 1 1 1 1 1 1 8 H+O2=O+OH 0 1 1 1 1 1 1 1 1 8 2H+M=H2+M 0 1 1 1 1 1 1 1 1 8 H+OH+M=H2O+M 0 1 1 1 1 1 1 1 1 8 H+HO2=O2+H2 0 1 1 1 1 1 1 1 1 8 H+HO2=2OH 0 1 1 1 1 1 1 1 1 8 H+CH3+M=CH4+M 0 1 1 1 1 1 1 1 1 8 H+CH4=CH3+H2 0 1 1 1 1 1 1 1 1 8 H+HCO=H2+CO 0 1 1 1 1 1 1 1 1 8 H+CH2O=HCO+H2 0 1 1 1 1 1 1 1 1 8 OH+H2=H+H2O 0 1 1 1 1 1 1 1 1 8 2OH=O+H2O 0 1 1 1 1 1 1 1 1 8 OH+HO2=O2+H2O 0 1 1 1 1 1 1 1 1 8 OH+CH4=CH3+H2O 0 1 1 1 1 1 1 1 1 8 OH+CO=H+CO2 0 1 1 1 1 1 1 1 1 8 OH+CH2O=HCO+H2O 0 1 1 1 1 1 1 1 1 8 HCO+M=H+CO+M 0 1 1 1 1 1 1 1 1 8 OH+CH3=CH2+H2O 0 0 1 1 1 1 1 1 1 7 CH3+O2=O+CH3O 0 1 1 0 1 1 1 1 1 7 O+H+M=OH+M 0 0 0 1 1 1 1 1 1 6 O+HO2=OH+O2 0 0 0 1 1 1 1 1 1 6 O+CH2=H+HCO 0 0 0 1 1 1 1 1 1 6 O+CH4=OH+CH3 0 0 0 1 1 1 1 1 1 6 O+CO+M=CO2+M 0 0 0 1 1 1 1 1 1 6 O+HCO=OH+CO 0 0 0 1 1 1 1 1 1 6 O+HCO=H+CO2 0 0 0 1 1 1 1 1 1 6 O+CH2O=OH+HCO 0 0 0 1 1 1 1 1 1 6 O2+CO=O+CO2 0 0 0 1 1 1 1 1 1 6 O2+CH2O=HO2+HCO 0 0 0 1 1 1 1 1 1 6 H+2O2=HO2+O2 0 0 0 1 1 1 1 1 1 6 H+O2+H2O=HO2+H2O 0 0 0 1 1 1 1 1 1 6 H+O2+N2=HO2+N2 0 0 0 1 1 1 1 1 1 6 2H+H2O=H2+H2O 0 0 0 1 1 1 1 1 1 6 2H+CO2=H2+CO2 0 0 0 1 1 1 1 1 1 6 H+CH2+M=CH3+M 0 0 0 1 1 1 1 1 1 6 H+HCO+M=CH2O+M 0 0 0 1 1 1 1 1 1 6 H2+CO+M=CH2O+M 0 0 0 1 1 1 1 1 1 6 OH+CH2=H+CH2O 0 0 0 1 1 1 1 1 1 6 OH+CH2(S)=H+CH2O 0 0 0 1 1 1 1 1 1 6 OH+CH3=CH2(S)+H2O 0 0 0 1 1 1 1 1 1 6 OH+HCO=H2O+CO 0 0 0 1 1 1 1 1 1 6 HO2+CH2=OH+CH2O 0 0 0 1 1 1 1 1 1 6 HO2+CH3=O2+CH4 0 0 0 1 1 1 1 1 1 6 HO2+CO=OH+CO2 0 0 0 1 1 1 1 1 1 6 CH2+H2=H+CH3 0 0 0 1 1 1 1 1 1 6 CH2+CH4=2CH3 0 0 0 1 1 1 1 1 1 6 CH2(S)+N2=CH2+N2 0 0 0 1 1 1 1 1 1 6 CH2(S)+O2=H+OH+CO 0 0 0 1 1 1 1 1 1 6 CH2(S)+O2=CO+H2O 0 0 0 1 1 1 1 1 1 6 CH2(S)+H2=CH3+H 0 0 0 1 1 1 1 1 1 6 CH2(S)+H2O=CH2+H2O 0 0 0 1 1 1 1 1 1 6 CH2(S)+CH4=2CH3 0 0 0 1 1 1 1 1 1 6 CH2(S)+CO=CH2+CO 0 0 0 1 1 1 1 1 1 6 CH2(S)+CO2=CH2+CO2 0 0 0 1 1 1 1 1 1 6 CH2(S)+CO2=CO+CH2O 0 0 0 1 1 1 1 1 1 6 CH3+O2=OH+CH2O 0 0 0 1 1 1 1 1 1 6 CH3+HCO=CH4+CO 0 0 0 1 1 1 1 1 1 6 CH3+CH2O=HCO+CH4 0 0 0 1 1 1 1 1 1 6 HCO+H2O=H+CO+H2O 0 0 0 1 1 1 1 1 1 6 HCO+O2=HO2+CO 0 0 0 1 1 1 1 1 1 6 2OH+M=H2O2+M 0 1 1 0 1 0 1 1 1 6 OH+H2O2=HO2+H2O 0 1 1 0 1 0 1 1 1 6 2HO2=O2+H2O2 0 1 1 0 1 0 1 1 1 6 O+CH2(S)=H+HCO 0 0 0 0 1 1 1 1 1 5 O+C2H4=CH3+HCO 0 0 0 0 1 1 1 1 1 5 O+C2H5=CH3+CH2O 0 0 0 0 1 1 1 1 1 5 O+C2H6=OH+C2H5 0 0 0 0 1 1 1 1 1 5 H+CH2O+M=CH3O+M 0 0 0 0 1 1 1 1 1 5 H+CH3O=OH+CH3 0 0 0 0 1 1 1 1 1 5 H+C2H4+M=C2H5+M 0 0 0 0 1 1 1 1 1 5 H+C2H5+M=C2H6+M 0 0 0 0 1 1 1 1 1 5 H+C2H6=C2H5+H2 0 0 0 0 1 1 1 1 1 5 OH+C2H6=C2H5+H2O 0 0 0 0 1 1 1 1 1 5 HO2+CH3=OH+CH3O 0 0 0 0 1 1 1 1 1 5 CH2+CH3=H+C2H4 0 0 0 0 1 1 1 1 1 5 CH2(S)+CH3=H+C2H4 0 0 0 0 1 1 1 1 1 5 2CH3+M=C2H6+M 0 0 0 0 1 1 1 1 1 5 2CH3=H+C2H5 0 0 0 0 1 1 1 1 1 5 CH3+C2H6=C2H5+CH4 0 0 0 0 1 1 1 1 1 5 CH3O+O2=HO2+CH2O 0 0 0 0 1 1 1 1 1 5 C2H5+O2=HO2+C2H4 0 0 0 0 1 1 1 1 1 5 CH3O+H=H2+CH2O 0 1 1 0 1 0 0 1 1 5 H+O2+AR=HO2+AR 0 0 0 0 0 1 1 1 1 4 CH2+O2=OH+HCO 0 0 0 1 0 1 1 1 0 4 CH2(S)+AR=CH2+AR 0 0 0 0 0 1 1 1 1 4 O+C2H2=CO+CH2 0 0 0 0 1 0 1 1 1 4 H+H2O2=HO2+H2 0 0 0 0 1 0 1 1 1 4 H+C2H2+M=C2H3+M 0 0 0 0 1 0 1 1 1 4 H+C2H3+M=C2H4+M 0 0 0 0 1 0 1 1 1 4 H+C2H3=H2+C2H2 0 0 0 0 1 0 1 1 1 4 H+C2H4=C2H3+H2 0 0 0 0 1 0 1 1 1 4 OH+C2H2=CH3+CO 0 0 0 0 1 0 1 1 1 4 OH+C2H3=H2O+C2H2 0 0 0 0 1 0 1 1 1 4 OH+C2H4=C2H3+H2O 0 0 0 0 1 0 1 1 1 4 HO2+CH2O=HCO+H2O2 0 0 0 0 1 0 1 1 1 4 2CH2=H2+C2H2 0 0 0 0 1 0 1 1 1 4 CH3+H2O2=HO2+CH4 0 0 0 0 1 0 1 1 1 4 CH3+C2H4=C2H3+CH4 0 0 0 0 1 0 1 1 1 4 C2H3+O2=HCO+CH2O 0 0 0 0 1 0 1 1 1 4 C2H4+M=H2+C2H2+M 0 0 0 0 1 0 1 1 1 4 2O+M=O2+M 0 0 0 1 1 0 0 1 1 4 HO2+H=O+H2O 0 0 0 1 1 0 0 1 1 4 CH+O=H+CO 0 0 1 0 1 0 0 1 1 4 CH+OH=H+HCO 0 0 1 0 1 0 0 1 1 4 CH+O2=O+HCO 0 0 1 0 1 0 0 1 1 4 CH+CO2=HCO+CO 0 0 1 0 1 0 0 1 1 4 CH2+OH=CH+H2O 0 0 1 0 1 0 0 1 1 4 CH2OH+H=H2+CH2O 0 0 0 1 1 0 0 1 1 4 CH2OH+H=OH+CH3 0 0 0 1 1 0 0 1 1 4 CH2OH+H=CH2(S)+H2O 0 0 0 1 1 0 0 1 1 4 CH2OH+O=OH+CH2O 0 0 0 1 1 0 0 1 1 4 CH2OH+OH=H2O+CH2O 0 0 0 1 1 0 0 1 1 4 CH2OH+O2=HO2+CH2O 0 0 0 1 1 0 0 1 1 4 O+CH2(S)=H2+CO 0 0 0 1 1 0 0 1 1 4 H+CH2O+M=CH2OH+M 0 0 0 1 1 0 0 1 1 4 H2O2+H=OH+H2O 0 0 0 0 1 0 0 1 1 3 H2O2+O=OH+HO2 0 0 0 0 1 0 0 1 1 3 C+OH=H+CO 0 0 0 0 1 0 0 1 1 3 C+O2=O+CO 0 0 0 0 1 0 0 1 1 3 CH+H=C+H2 0 0 0 0 1 0 0 1 1 3 CH+H2=H+CH2 0 0 0 0 1 0 0 1 1 3 CH+H2O=H+CH2O 0 0 0 0 1 0 0 1 1 3 CH+CO+M=HCCO+M 0 0 0 0 1 0 0 1 1 3 CH2+CH=H+C2H2 0 0 0 0 1 0 0 1 1 3 CH2(S)+H=CH+H2 0 0 0 0 1 0 0 1 1 3 CH2(S)+H2O+M=CH3OH+M 0 0 0 0 1 0 0 1 1 3 CH2O+CH=H+CH2CO 0 0 0 0 1 0 0 1 1 3 CH3+OH+M=CH3OH+M 0 0 0 0 1 0 0 1 1 3 CH3+C=H+C2H2 0 0 0 0 1 0 0 1 1 3 CH3+CH=H+C2H3 0 0 0 0 1 0 0 1 1 3 CH3O+H+M=CH3OH+M 0 0 0 0 1 0 0 1 1 3 CH3O+H=H+CH2OH 0 0 0 0 1 0 0 1 1 3 CH3O+H=CH2(S)+H2O 0 0 0 0 1 0 0 1 1 3 CH3O+O=OH+CH2O 0 0 0 0 1 0 0 1 1 3 CH3O+OH=H2O+CH2O 0 0 0 0 1 0 0 1 1 3 CH2OH+H+M=CH3OH+M 0 0 0 0 1 0 0 1 1 3 CH4+CH=H+C2H4 0 0 0 0 1 0 0 1 1 3 CH3OH+H=CH2OH+H2 0 0 0 0 1 0 0 1 1 3 CH3OH+H=CH3O+H2 0 0 0 0 1 0 0 1 1 3 CH3OH+O=OH+CH2OH 0 0 0 0 1 0 0 1 1 3 CH3OH+O=OH+CH3O 0 0 0 0 1 0 0 1 1 3 CH3OH+OH=CH2OH+H2O 0 0 0 0 1 0 0 1 1 3 CH3OH+OH=CH3O+H2O 0 0 0 0 1 0 0 1 1 3 CH3OH+CH3=CH2OH+CH4 0 0 0 0 1 0 0 1 1 3 CH3OH+CH3=CH3O+CH4 0 0 0 0 1 0 0 1 1 3 HCCO+H=CH2(S)+CO 0 0 0 0 1 0 0 1 1 3 HCCO+O=H+2CO 0 0 0 0 1 0 0 1 1 3 HCCO+O2=OH+2CO 0 0 0 0 1 0 0 1 1 3 HCCO+CH=CO+C2H2 0 0 0 0 1 0 0 1 1 3 HCCO+CH2=C2H3+CO 0 0 0 0 1 0 0 1 1 3 2HCCO=2CO+C2H2 0 0 0 0 1 0 0 1 1 3 C2H2+O=H+HCCO 0 0 0 0 1 0 0 1 1 3 C2H2+OH=H+CH2CO 0 0 0 0 1 0 0 1 1 3 CH2+CO+M=CH2CO+M 0 0 0 0 1 0 0 1 1 3 CH2CO+H=HCCO+H2 0 0 0 0 1 0 0 1 1 3 CH2CO+H=CH3+CO 0 0 0 0 1 0 0 1 1 3 CH2CO+O=OH+HCCO 0 0 0 0 1 0 0 1 1 3 CH2CO+O=CH2+CO2 0 0 0 0 1 0 0 1 1 3 CH2CO+OH=HCCO+H2O 0 0 0 0 1 0 0 1 1 3 C2H3+O=H+CH2CO 0 0 0 0 1 0 0 1 1 3 C2H5+H=H2+C2H4 0 0 0 0 1 0 0 1 1 3 C2H6+CH2(S)=CH3+C2H5 0 0 0 0 1 0 0 1 1 3 H2O2+M=2OH+M 0 1 1 0 0 0 0 0 0 2 CH+H2+M=CH3+M 0 0 0 0 1 0 0 0 1 2 CH2+O2=OH+H+CO 0 0 0 0 1 0 0 0 1 2 CH2+O2=2H+CO2 0 0 0 0 1 0 0 0 1 2 CH2+O2=O+CH2O 0 0 0 0 1 0 0 0 1 2 CH2+C=H+C2H 0 0 0 0 0 0 0 1 1 2 2CH2=2H+C2H2 0 0 0 0 1 0 0 0 1 2 CH2(S)+H2O=H2+CH2O 0 0 0 0 1 0 0 0 1 2 CH3+O=H+H2+CO 0 0 0 0 1 0 0 0 1 2 CH3+OH=H2+CH2O 0 0 0 0 1 0 0 0 1 2 CH3O+M=H+CH2O+M 0 1 1 0 0 0 0 0 0 2 C2H+H+M=C2H2+M 0 0 0 0 0 0 0 1 1 2 C2H+O=CH+CO 0 0 0 0 0 0 0 1 1 2 C2H+OH=H+HCCO 0 0 0 0 0 0 0 1 1 2 C2H+H2=H+C2H2 0 0 0 0 0 0 0 1 1 2 C2H+O2=HCO+CO 0 0 0 0 0 0 0 1 1 2 C2H2+OH=C2H+H2O 0 0 0 0 0 0 0 1 1 2 C2H3+O2=CH2CHO+O 0 0 0 0 1 0 0 0 1 2 C2H3+O2=C2H2+HO2 0 0 0 0 1 0 0 0 1 2 CH2CHO+H=CH3+HCO 0 0 0 0 1 0 0 0 1 2 CH2CHO+H=CH2CO+H2 0 0 0 0 1 0 0 0 1 2 CH2CHO+O=H+CH2+CO2 0 0 0 0 1 0 0 0 1 2 CH2CHO+OH=H2O+CH2CO 0 0 0 0 1 0 0 0 1 2 CH2CHO+OH=HCO+CH2OH 0 0 0 0 1 0 0 0 1 2 CH2CHO+O2=OH+CO+CH2O 0 0 0 0 1 0 0 0 1 2 C2H4+O=H+CH2CHO 0 0 0 0 1 0 0 0 1 2 O+C2H2=OH+C2H 0 0 0 0 0 0 0 1 1 2 H+HCCOH=H+CH2CO 0 0 0 0 0 0 0 1 1 2 OH+C2H2=H+HCCOH 0 0 0 0 0 0 0 1 1 2 H+CH2CO+M=CH2CHO+M 0 0 0 0 1 0 0 0 1 2 O2+CH2CHO=OH+2HCO 0 0 0 0 1 0 0 0 1 2 O+OH=O2+H 0 1 1 0 0 0 0 0 0 2 OH+H=O+H2 0 1 1 0 0 0 0 0 0 2 H2O+H=H2+OH 0 1 1 0 0 0 0 0 0 2 H2O+O=OH+OH 0 1 1 0 0 0 0 0 0 2 CO2+H=CO+OH 0 1 1 0 0 0 0 0 0 2 CH4=CH3+H 0 1 1 0 0 0 0 0 0 2 CH3+H2=CH4+H 0 1 1 0 0 0 0 0 0 2 CH3+H2O=CH4+OH 0 1 1 0 0 0 0 0 0 2 H2O+HO2=H2O2+OH 0 1 1 0 0 0 0 0 0 2 O+C2H2=CH2(S)+CO 0 0 0 0 0 0 1 0 0 1 CH3CHO+H=CH2CHO+H2 0 0 0 0 0 0 0 0 1 1 CH3CHO+O=OH+CH2CHO 0 0 0 0 0 0 0 0 1 1 C2H5+O=H+CH3CHO 0 0 0 0 0 0 0 0 1 1 N+NO=N2+O 0 0 0 0 0 0 0 0 1 1 N+O2=NO+O 0 0 0 0 0 0 0 0 1 1 N+OH=NO+H 0 0 0 0 0 0 0 0 1 1 N2O+O=N2+O2 0 0 0 0 0 0 0 0 1 1 N2O+O=2NO 0 0 0 0 0 0 0 0 1 1 N2O+H=N2+OH 0 0 0 0 0 0 0 0 1 1 N2O+OH=N2+HO2 0 0 0 0 0 0 0 0 1 1 N2O+M=N2+O+M 0 0 0 0 0 0 0 0 1 1 HO2+NO=NO2+OH 0 0 0 0 0 0 0 0 1 1 NO+O+M=NO2+M 0 0 0 0 0 0 0 0 1 1 NO2+O=NO+O2 0 0 0 0 0 0 0 0 1 1 NO2+H=NO+OH 0 0 0 0 0 0 0 0 1 1 NH+O=NO+H 0 0 0 0 0 0 0 0 1 1 NH+H=N+H2 0 0 0 0 0 0 0 0 1 1 NH+OH=HNO+H 0 0 0 0 0 0 0 0 1 1 NH+OH=N+H2O 0 0 0 0 0 0 0 0 1 1 NH+O2=HNO+O 0 0 0 0 0 0 0 0 1 1 NH+O2=NO+OH 0 0 0 0 0 0 0 0 1 1 NH+N=N2+H 0 0 0 0 0 0 0 0 1 1 NH+H2O=HNO+H2 0 0 0 0 0 0 0 0 1 1 NH+NO=N2+OH 0 0 0 0 0 0 0 0 1 1 NH+NO=N2O+H 0 0 0 0 0 0 0 0 1 1 NH2+O=OH+NH 0 0 0 0 0 0 0 0 1 1 NH2+O=H+HNO 0 0 0 0 0 0 0 0 1 1 NH2+H=NH+H2 0 0 0 0 0 0 0 0 1 1 NH2+OH=NH+H2O 0 0 0 0 0 0 0 0 1 1 NNH=N2+H 0 0 0 0 0 0 0 0 1 1 NNH+O2=HO2+N2 0 0 0 0 0 0 0 0 1 1 NNH+O=OH+N2 0 0 0 0 0 0 0 0 1 1 NNH+O=NH+NO 0 0 0 0 0 0 0 0 1 1 NNH+H=H2+N2 0 0 0 0 0 0 0 0 1 1 NNH+OH=H2O+N2 0 0 0 0 0 0 0 0 1 1 NNH+CH3=CH4+N2 0 0 0 0 0 0 0 0 1 1 H+NO+M=HNO+M 0 0 0 0 0 0 0 0 1 1 HNO+O=NO+OH 0 0 0 0 0 0 0 0 1 1 HNO+H=H2+NO 0 0 0 0 0 0 0 0 1 1 HNO+OH=NO+H2O 0 0 0 0 0 0 0 0 1 1 HNO+O2=HO2+NO 0 0 0 0 0 0 0 0 1 1 CN+O=CO+N 0 0 0 0 0 0 0 0 1 1 CN+OH=NCO+H 0 0 0 0 0 0 0 0 1 1 CN+H2O=HCN+OH 0 0 0 0 0 0 0 0 1 1 CN+O2=NCO+O 0 0 0 0 0 0 0 0 1 1 CN+H2=HCN+H 0 0 0 0 0 0 0 0 1 1 NCO+O=NO+CO 0 0 0 0 0 0 0 0 1 1 NCO+H=NH+CO 0 0 0 0 0 0 0 0 1 1 NCO+OH=NO+H+CO 0 0 0 0 0 0 0 0 1 1 NCO+N=N2+CO 0 0 0 0 0 0 0 0 1 1 NCO+O2=NO+CO2 0 0 0 0 0 0 0 0 1 1 NCO+M=N+CO+M 0 0 0 0 0 0 0 0 1 1 NCO+NO=N2O+CO 0 0 0 0 0 0 0 0 1 1 NCO+NO=N2+CO2 0 0 0 0 0 0 0 0 1 1 HCN+M=H+CN+M 0 0 0 0 0 0 0 0 1 1 HCN+O=NCO+H 0 0 0 0 0 0 0 0 1 1 HCN+O=NH+CO 0 0 0 0 0 0 0 0 1 1 HCN+O=CN+OH 0 0 0 0 0 0 0 0 1 1 HCN+OH=HOCN+H 0 0 0 0 0 0 0 0 1 1 HCN+OH=HNCO+H 0 0 0 0 0 0 0 0 1 1 HCN+OH=NH2+CO 0 0 0 0 0 0 0 0 1 1 H+HCN+M=H2CN+M 0 0 0 0 0 0 0 0 1 1 H2CN+N=N2+CH2 0 0 0 0 0 0 0 0 1 1 C+N2=CN+N 0 0 0 0 0 0 0 0 1 1 CH+N2=HCN+N 0 0 0 0 0 0 0 0 1 1 CH+N2+M=HCNN+M 0 0 0 0 0 0 0 0 1 1 CH2+N2=HCN+NH 0 0 0 0 0 0 0 0 1 1 CH2(S)+N2=NH+HCN 0 0 0 0 0 0 0 0 1 1 C+NO=CN+O 0 0 0 0 0 0 0 0 1 1 C+NO=CO+N 0 0 0 0 0 0 0 0 1 1 CH+NO=HCN+O 0 0 0 0 0 0 0 0 1 1 CH+NO=H+NCO 0 0 0 0 0 0 0 0 1 1 CH+NO=N+HCO 0 0 0 0 0 0 0 0 1 1 CH2+NO=H+HNCO 0 0 0 0 0 0 0 0 1 1 CH2+NO=OH+HCN 0 0 0 0 0 0 0 0 1 1 CH2+NO=H+HCNO 0 0 0 0 0 0 0 0 1 1 CH2(S)+NO=H+HNCO 0 0 0 0 0 0 0 0 1 1 CH2(S)+NO=OH+HCN 0 0 0 0 0 0 0 0 1 1 CH2(S)+NO=H+HCNO 0 0 0 0 0 0 0 0 1 1 CH3+NO=HCN+H2O 0 0 0 0 0 0 0 0 1 1 CH3+NO=H2CN+OH 0 0 0 0 0 0 0 0 1 1 HCNN+O=CO+H+N2 0 0 0 0 0 0 0 0 1 1 HCNN+O=HCN+NO 0 0 0 0 0 0 0 0 1 1 HCNN+O2=O+HCO+N2 0 0 0 0 0 0 0 0 1 1 HCNN+OH=H+HCO+N2 0 0 0 0 0 0 0 0 1 1 HCNN+H=CH2+N2 0 0 0 0 0 0 0 0 1 1 HNCO+O=NH+CO2 0 0 0 0 0 0 0 0 1 1 HNCO+O=HNO+CO 0 0 0 0 0 0 0 0 1 1 HNCO+O=NCO+OH 0 0 0 0 0 0 0 0 1 1 HNCO+H=NH2+CO 0 0 0 0 0 0 0 0 1 1 HNCO+H=H2+NCO 0 0 0 0 0 0 0 0 1 1 HNCO+OH=NCO+H2O 0 0 0 0 0 0 0 0 1 1 HNCO+OH=NH2+CO2 0 0 0 0 0 0 0 0 1 1 HNCO+M=NH+CO+M 0 0 0 0 0 0 0 0 1 1 HCNO+H=H+HNCO 0 0 0 0 0 0 0 0 1 1 HCNO+H=OH+HCN 0 0 0 0 0 0 0 0 1 1 HCNO+H=NH2+CO 0 0 0 0 0 0 0 0 1 1 HOCN+H=H+HNCO 0 0 0 0 0 0 0 0 1 1 HCCO+NO=HCNO+CO 0 0 0 0 0 0 0 0 1 1 CH3+N=H2CN+H 0 0 0 0 0 0 0 0 1 1 CH3+N=HCN+H2 0 0 0 0 0 0 0 0 1 1 NH3+H=NH2+H2 0 0 0 0 0 0 0 0 1 1 NH3+OH=NH2+H2O 0 0 0 0 0 0 0 0 1 1 NH3+O=NH2+OH 0 0 0 0 0 0 0 0 1 1 NH+CO2=HNO+CO 0 0 0 0 0 0 0 0 1 1 CN+NO2=NCO+NO 0 0 0 0 0 0 0 0 1 1 NCO+NO2=N2O+CO2 0 0 0 0 0 0 0 0 1 1 N+CO2=NO+CO 0 0 0 0 0 0 0 0 1 1 O+CH3CHO=OH+CH3+CO 0 0 0 0 0 0 0 0 1 1 O2+CH3CHO=HO2+CH3+CO 0 0 0 0 0 0 0 0 1 1 H+CH3CHO=CH3+H2+CO 0 0 0 0 0 0 0 0 1 1 OH+CH3CHO=CH3+H2O+CO 0 0 0 0 0 0 0 0 1 1 HO2+CH3CHO=CH3+H2O2+CO 0 0 0 0 0 0 0 0 1 1 CH3+CH3CHO=CH3+CH4+CO 0 0 0 0 0 0 0 0 1 1 CH3+C2H5+M=C3H8+M 0 0 0 0 0 0 0 0 1 1 O+C3H8=OH+C3H7 0 0 0 0 0 0 0 0 1 1 H+C3H8=C3H7+H2 0 0 0 0 0 0 0 0 1 1 OH+C3H8=C3H7+H2O 0 0 0 0 0 0 0 0 1 1 C3H7+H2O2=HO2+C3H8 0 0 0 0 0 0 0 0 1 1 CH3+C3H8=C3H7+CH4 0 0 0 0 0 0 0 0 1 1 CH3+C2H4+M=C3H7+M 0 0 0 0 0 0 0 0 1 1 O+C3H7=C2H5+CH2O 0 0 0 0 0 0 0 0 1 1 H+C3H7+M=C3H8+M 0 0 0 0 0 0 0 0 1 1 H+C3H7=CH3+C2H5 0 0 0 0 0 0 0 0 1 1 OH+C3H7=C2H5+CH2OH 0 0 0 0 0 0 0 0 1 1 HO2+C3H7=O2+C3H8 0 0 0 0 0 0 0 0 1 1 HO2+C3H7=OH+C2H5+CH2O 0 0 0 0 0 0 0 0 1 1 CH3+C3H7=2C2H5 0 0 0 0 0 0 0 0 1 1 H2+0.5O2=1H2O 1 0 0 0 0 0 0 0 0 1 H2O+CO=H2+CO2 1 0 0 0 0 0 0 0 0 1 CH4+H2O=1CO+3H2 1 0 0 0 0 0 0 0 0 1 CH4+0.5O2=1CO+2H2 1 0 0 0 0 0 0 0 0 1 CH2+O=CO+H2 0 0 1 0 0 0 0 0 0 1 注:1为包含,0为不包含。 -
[1] 张全. 简化燃烧化学反应机理的研究[D]. 合肥: 中国科学技术大学, 2001.ZHANG Q. Simplified study of combustion chemical reaction mechanism[D]. Hefei: University of Science and Technology of China, 2001(in Chinese). [2] 谭洵. 甲烷扩散火焰的富氧燃烧特性及总包反应机理研究[D]. 武汉: 华中科技大学, 2014.TAN X. Oxy-fuel combustion characteristics and the global chemistry mechanism for methane diffusion flame[D]. Wuhan: Huazhong University of Science and Technology, 2014(in Chinese). [3] 林志勇, 周进, 黄玉辉. 基于敏感性分析和准稳态假设简化详细反应机理[J]. 国防科技大学学报, 2007, 29(1): 16-20.LIN Z Y, ZHOU J, HUANG Y H. Simplification of detailed reaction mechanism based on the sensitivity analysis and quasi-steady state assumption[J]. Journal of National University of Defense Technology, 2007, 29(1): 16-20(in Chinese). [4] FOMIN A, ZAVLEV T, ALEKSEEV V A, et al. Experimental and modelling study of 1CH2 in premixed very rich methane flames[J]. Combustion and Flame, 2016, 171: 198-210. [5] SATIJA A, YUAN S L, NAIK S V, et al. Vibrational CARS thermometry and one-dimensional numerical simulations in CH4/H2/air partially-premixed flames[J]. International Journal of Hydrogen Energy, 2015, 40(21): 6959-6969. [6] PILLIER L, IDIR M, MOLET J, et al. Experimental study and modelling of NOx formation in high pressure counter-flow premixed CH4/air flames[J]. Fuel, 2015, 150: 394-407. [7] HE D, YU Y S, KUANG Y C, et al. Model comparisons of flow and chemical kinetic mechanisms for methane–air combustion for engineering applications[J]. Applied Sciences, 2021, 11(9): 4107 . [8] 董刚, 黄鹰, 陈义良. 不同化学反应机理对甲烷射流湍流扩散火焰计算结果影响的研究[J]. 燃料化学学报, 2000, 28(1): 49-54.DONG G, HUANG Y, CHEN Y L. Study of effects of different chemical reaction mechanisms on computation results for methane jet turbulence diffusion flame[J]. Journal of Fuel Chemistry and Technology, 2000, 28(1): 49-54(in Chinese). [9] BARLOW R S, FRANK J H. Effects of turbulence on species mass fractions in methane/air jet flames[J]. Symposium (International) on Combustion, 1998, 27(1): 1087-1095. [10] 宋绪光, 金捷, 张敏琦, 等. 基于LES的射流火焰湍流辐射交互作用研究[J]. 北京航空航天大学学报, 2024, 50(8): 2667-2676.SONG X G, JIN J, ZHANG M Q, et al. Turbulence-radiation interaction in turbulent jet flame based on large-eddy simulation[J]. Journal of Beijing University of Aeronautics and Astronautics, 2024, 50(8): 2667-2676(in Chinese). [11] JONES W P, LINDSTEDT R P. Global reaction schemes for hydrocarbon combustion[J]. Combustion and Flame, 1988, 73(3): 233-249. [12] CHANG W C, CHEN J Y. Reduced mechanisms based on GRI-Mech 1.2 [EB/OL]. (2020-01-22)[2023-01-22]. http://firebrand.me.berkeley.edu/reduced. [13] SUNG C J, LAW C K, CHEN J Y. Augmented reduced mechanisms for NO emission in methane oxidation[J]. Combustion and Flame, 2001, 125(1-2): 906-919. [14] SANKARAN R, HAWKES E R, CHEN J H, et al. Structure of a spatially developing turbulent lean methane–air Bunsen flame[J]. Proceedings of the Combustion Institute, 2007, 31(1): 1291-1298. [15] LU T F, LAW C K. A criterion based on computational singular perturbation for the identification of quasi steady state species: a reduced mechanism for methane oxidation with NO chemistry[J]. Combustion and Flame, 2008, 154(4): 761-774. [16] SMOOKE M D. Reduced kinetic mechanisms and asymptotic approximations for methane-air flames[M]. Berlin: Springer-Verlag, 1991. [17] LARSSON A, ZETTERVALL N, HURTIG T, et al. Skeletal methane–air reaction mechanism for large eddy simulation of turbulent microwave-assisted combustion[J]. Energy & Fuels, 2017, 31(2): 1904-1926. [18] Kazakov A, Frenklach M. Reduced reaction sets based on GRI-mech 1.2 [EB/OL]. (2023-11-19)[2023-12-20]. http://www.me.berkeley.edu/drm/. [19] MICHAEL F. GRI-Mech home page[EB/OL]. (2021-03-01)[ 2023-12-20]. http://combustion.berkeley.edu/gri-mech. [20] LAW C K, MAKINO A, LU T F. On the off-stoichiometric peaking of adiabatic flame temperature[J]. Combustion and Flame, 2006, 145(4): 808-819. [21] PARK O, VELOO P S, LIU N, et al. Combustion characteristics of alternative gaseous fuels[J]. Proceedings of the Combustion Institute, 2011, 33(1): 887-894. [22] HU E J, LI X T, MENG X, et al. Laminar flame speeds and ignition delay times of methane–air mixtures at elevated temperatures and pressures[J]. Fuel, 2015, 158: 1-10. -





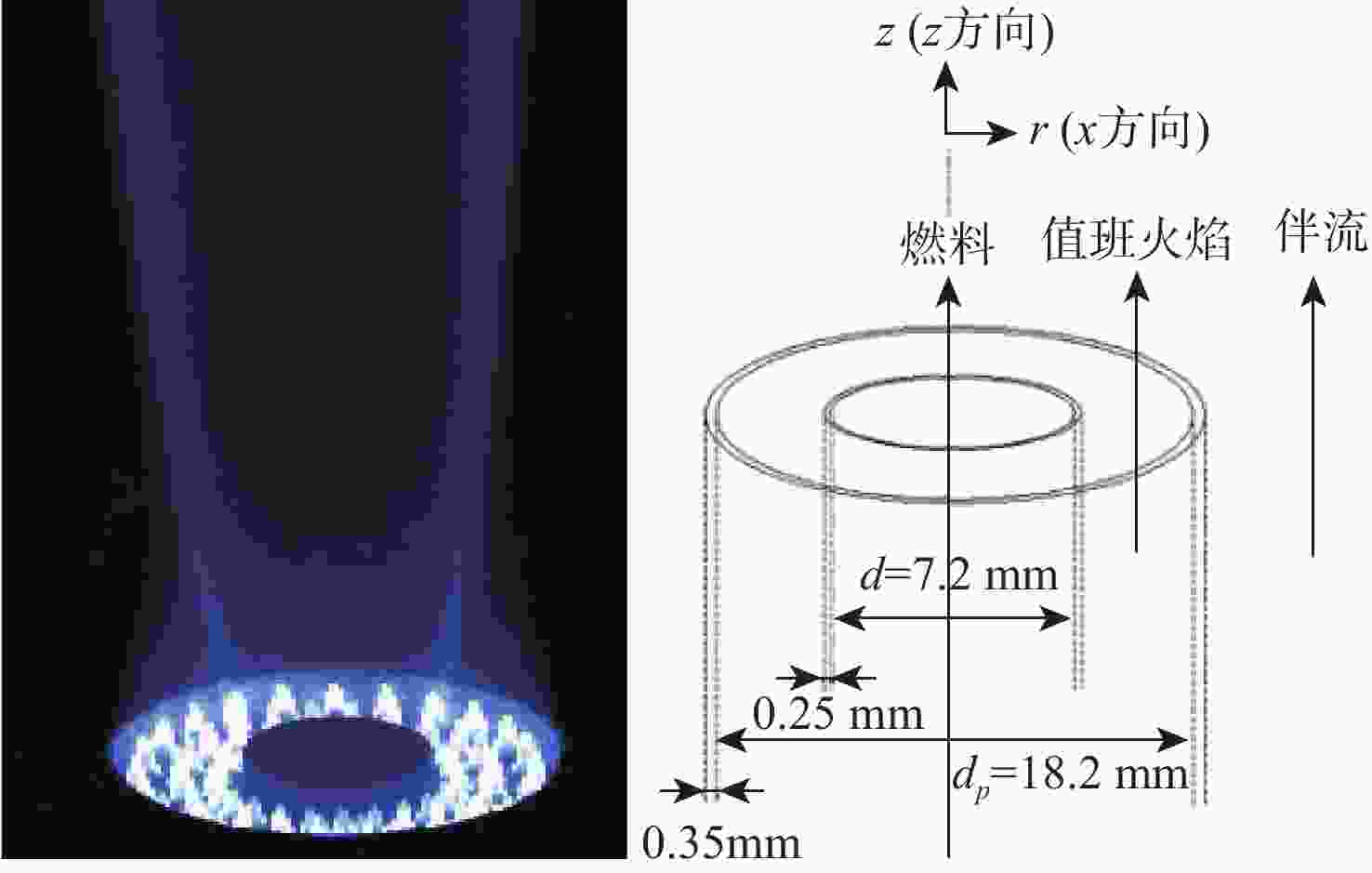
 下载:
下载: